The rapid growth of biotechnology has revolutionized healthcare, agriculture, and environmental research, while simultaneously reshaping the landscape of intellectual property rights (IPR). Companies working in areas like genetic engineering, precision medicine, and diagnostic innovation operate in a highly competitive, research-driven environment. In such a scenario, protecting intellectual property is critical for sustaining innovation, attracting investment, and maintaining a competitive edge.
In India, biotechnology firms rely on a multi-layered approach to protect their innovations. This strategy integrates patents, trade secrets, contractual agreements and regulatory compliance, creating a robust system that secures both scientific discoveries and commercial advantages.
Patent Protection: The Foundation of IP Strategy
Patents form the cornerstone of intellectual property protection. Indian biotech companies typically pursue patents for innovations such as:
- engineered genetic constructs and recombinant proteins,
- novel microorganisms and cell lines, and
- diagnostic kits, reagents, and laboratory devices.
The graph below shows a steady rise in biotechnology patent applications filed at the Indian Patent Office since the early 1990s. Starting from just one application in 1991, filings grew significantly after 2000, driven by increased IP awareness, R&D efforts, and commercialization in India’s biotech sector.
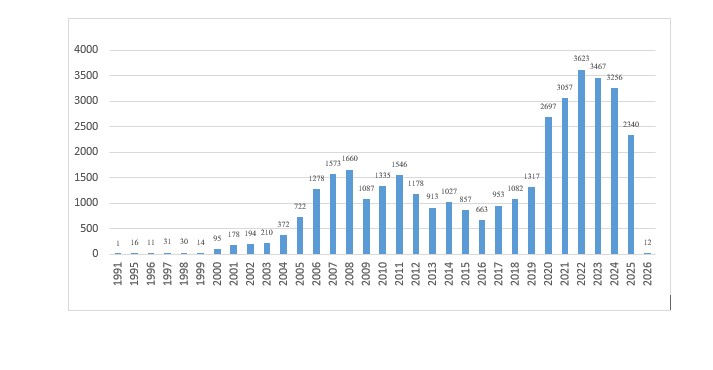
For certain inventions, depositing biological material with an authorized repository under the Budapest Treaty may be required. Additionally, when research involves India’s native biological resources, approval from the National Biodiversity Authority ensures compliance with ethical standards and benefit-sharing obligations.
International Protection and Enforcement
Many Indian biotech firms pursue global patent coverage, filing in jurisdictions such as the United States, Europe, Japan and the like. Treaties/ provisions such as the Patent Cooperation Treaty (PCT) provide broad territorial protection and strengthen investor confidence. International patents also improve a company’s leverage in licensing and collaborative agreements.
Enforcement is equally important and companies can:
- initiate civil litigation in Indian courts,
- seek injunctions and claim damages, and
- work with customs authorities to prevent unauthorized imports.
While legal proceedings can be lengthy, having enforceable remedies act as a strong deterrent against infringement.
Contractual Measures and Confidentiality
Patents alone are not enough to protect sensitive biotechnology innovations. Contractual safeguards play a crucial role, especially during collaborations with universities, research institutions, or international partners. Key tools include:
- non-disclosure agreements (NDAs): To prevent unauthorized sharing of proprietary research, laboratory protocols, or genomic information.
- employment contracts: Including confidentiality clauses and invention assignment agreements to ensure innovations developed by employees are legally owned by the company.
These agreements/ contracts are particularly valuable during early-stage research, before patent applications are filed.
Trade Secrets
Trade secrets provide an additional layer of protection, particularly for methods and processes that are difficult to patent or reverse engineer. In India, trade secret protection is primarily enforced through contract law and equitable principles.
Biotech companies often rely on trade secrets for:
- proprietary algorithms and bioinformatics tools,
- manufacturing techniques and cell culture protocols, and
- curated genetic databases and data analytics methods.
In diagnostic development, software and analytical databases often carry more commercial value than the physical test kits themselves. Hence, strict internal controls, restricted access, and robust digital security protocols are essential to maintain the confidentiality of these assets.
Trademarks and Copyrights
Branding and digital assets also require protection.
- trademarks: secure product names, logos, and identifiers associated with diagnostic tests or genetic services, helping prevent market confusion.
- copyrights: Protect software code, training manuals, laboratory documentation, and analytical tools.
With the increasing role of software in precision medicine, copyright protection of bioinformatics platforms is becoming increasingly critical.
Regulatory Alignment
Intellectual property strategies must align with regulatory requirements to ensure protection without compromising disclosures. Diagnostic kits and in vitro devices must comply with standards set by the Central Drugs Standard Control Organization (CDSCO). Timely patent filings to precede regulatory approvals helps preserve both novelty and enforceability.
Government Support
Government initiatives play a critical role in reinforcing innovation protection. National biotechnology policies encourage commercialization while emphasizing biosafety, ethical governance, and sustainability. Funding agencies often require detailed IP management plans, promoting structured strategies from the start of research projects.
Conclusion
Indian biotechnology companies protect genetic and diagnostic innovations through a comprehensive, multi-tiered approach:
- patents safeguard scientific inventions,
- contracts and trade secrets preserve proprietary knowledge,
- trademarks and copyrights protect brand identity and digital assets,
- regulatory compliance ensures commercial integrity, and
- international filings expand global reach.
This integrated framework creates a resilient ecosystem that balances innovation, ethical responsibility, and economic growth, allowing India’s biotech sector to thrive both domestically and on the global stage.


